
Presentation of the divison
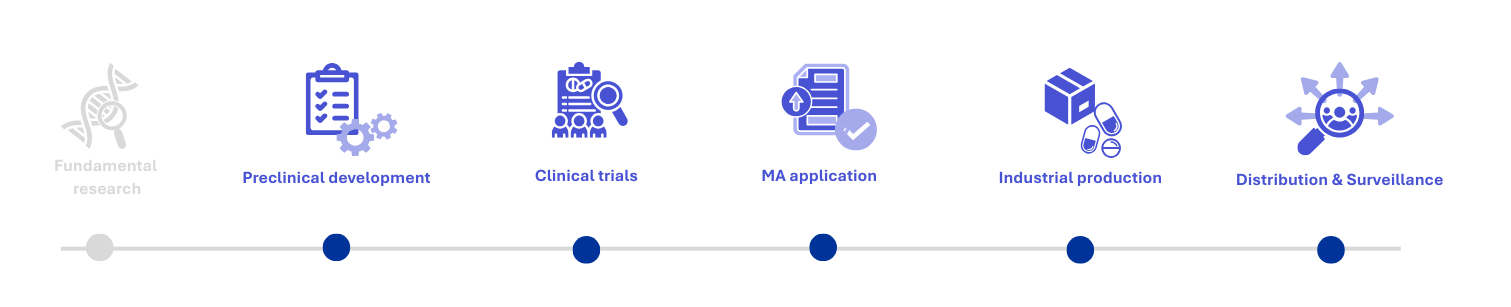
Working alongside you, we support the strategic development of your pharmaceutical products, cosmetic products, and medical devices. Our teams are made up of experts who are able to support you through every aspect of the life cycle of your products: clinical trials, early access authorisations, registration, CE marking, and post-MA activities (product information updates, advertising, etc.).

Our expertise – Regulatory affairs
Advertising Compliance Officer
CMC Technical Regulatory Writing Expert
Submissions and Registration Specialist
Global Regulatory Affairs Consultant
FULL PRODUCT LIFECYCLE SUPPORT
Regulatory Affairs
Our expertise – Vigilance & Safety
PV Case Processing Manager
Cosmetovigilance Officer / Toxicologist
Safety Database Administrator
Signal Detection and Risk Management Expert
Periodic Safety Report Writer (Safety Report Lead)
PV Auditor (QA / PV)
Qualified Person for Pharmacovigilance (EU-QPPV)
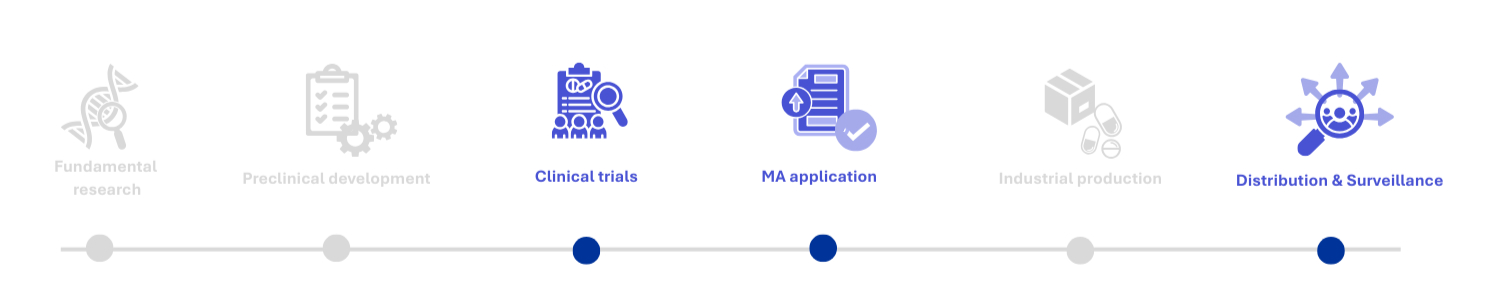
FULL PRODUCT LIFECYCLE SUPPORT
Vigilance
Therapeutic Areas
- Pharmaceuticals
- Biotech
- Medical Devices
- Healthcare Facilities
- Cosmetics
- Nutrition
- Animal Health
Industries
-
Oncology & Hematology
-
Vaccines
-
Rare Diseases
-
Pulmonology
-
Immunology
-
Dermatology
-
Neuroscience
-
Cardio-metabolism
-
Allergology
-
Ophthalmology
Our Achievements






